How is Coulter Different from Other Grants?
Coulter funds translational research projects that have the potential to improve patient and health care. Awards of up to $100,000 for a year are used for product definition studies (e.g. feasibility studies, prototype development, proof-of-concept studies) comprised of critical go/no-go milestones. Applicants who are invited to submit a full proposal work with the Coulter Program Office and/or mentors to validate their unmet medical need, and establish a technology development and commercialization plan. Funded projects are actively managed by the Program Office under a market-focused project management oversight and decision-making plan. Continued funding of a Coulter project is contingent upon successful and timely completion of project milestones.
Full Bridge Grant vs. Seed Grant Requests
The MU Coulter Program supports two kinds of project funding: full and seed. Full proposals are for projects that require up to $100K to complete experiments that validate the technology or kill the project. Full grants fund projects for up to one year. Full projects should be 12-24 months from a commercial event like a license to an established company or funding sufficient to continue translation of the technology. Seed grants are for projects that are earlier in nature and/or require $25K or less to complete major milestones and qualify for full applications. MU Coulter Seed Grant proposals are accepted all year around, however, funding may be contingent upon successful completion of Coulter Boot Camp.
Call for LOIs for the 2021 Coulter Grants will go out in November 2020, and the deadline for LOI submission will be Monday, December 21, 2020.
Eligibility
Investigator: All projects must include an engineer and a clinician who serve as Co-Principal Investigators (Co-PIs). All Co-PIs must be from the MU (Columbia) campus. Collaborators from other UM System campuses and from other universities may participate, however, award funding may not be used for research on other campuses. The engineer Co-PI must have a primary or joint appointment in the College of Engineering. The clinical Co-PI must have a primary or joint appointment in the School of Medicine and have job responsibilities that include direct contact with patients or patient data. Projects may include more than one engineering Co-PI and more than one clinical Co-PI as well as collaborators from the School of Nursing, College of Veterinary Medicine or allied health professionals. Exceptions to these criteria will be considered on a case by case basis. A Co-PI may submit more than one LOI/proposal; however, it is rare for the same project team to receive funding for multiple projects in one funding cycle.
Product Type: The following types of products in the human health space are eligible: diagnostic, device, tool and software.
Intellectual Property Requirement: Applications should be based on ideas that originate from within MU (making them potentially eligible for intellectual property protection) or university technologies with pending/issued patents or copyright. Technologies that are already licensed or optioned to a company are not eligible.
Department Commitment
To receive Coulter funding, the Co-PIs' departments will agree to share 50% of any future royalties the departments may receive from technology licenses resulting from the Coulter Accelerator Program support. These funds will flow back to the Coulter Accelerator Program, providing a return on investment for the units that have provided the upfront investments in the Program.
Proposal Evaluation
The research must have a direct application to an important unmet clinical need and the proposed solution, when commercialized, must have a benefit to patients and community health. Proposals are evaluated on the basis of scientific and clinical merit, potential health care impact and significance, experience of the investigators, timeline and pathway to commercialization as well as the likelihood of obtaining the additional funding required to fully translate the proposed solution.
Coulter Application Guidelines and Steps
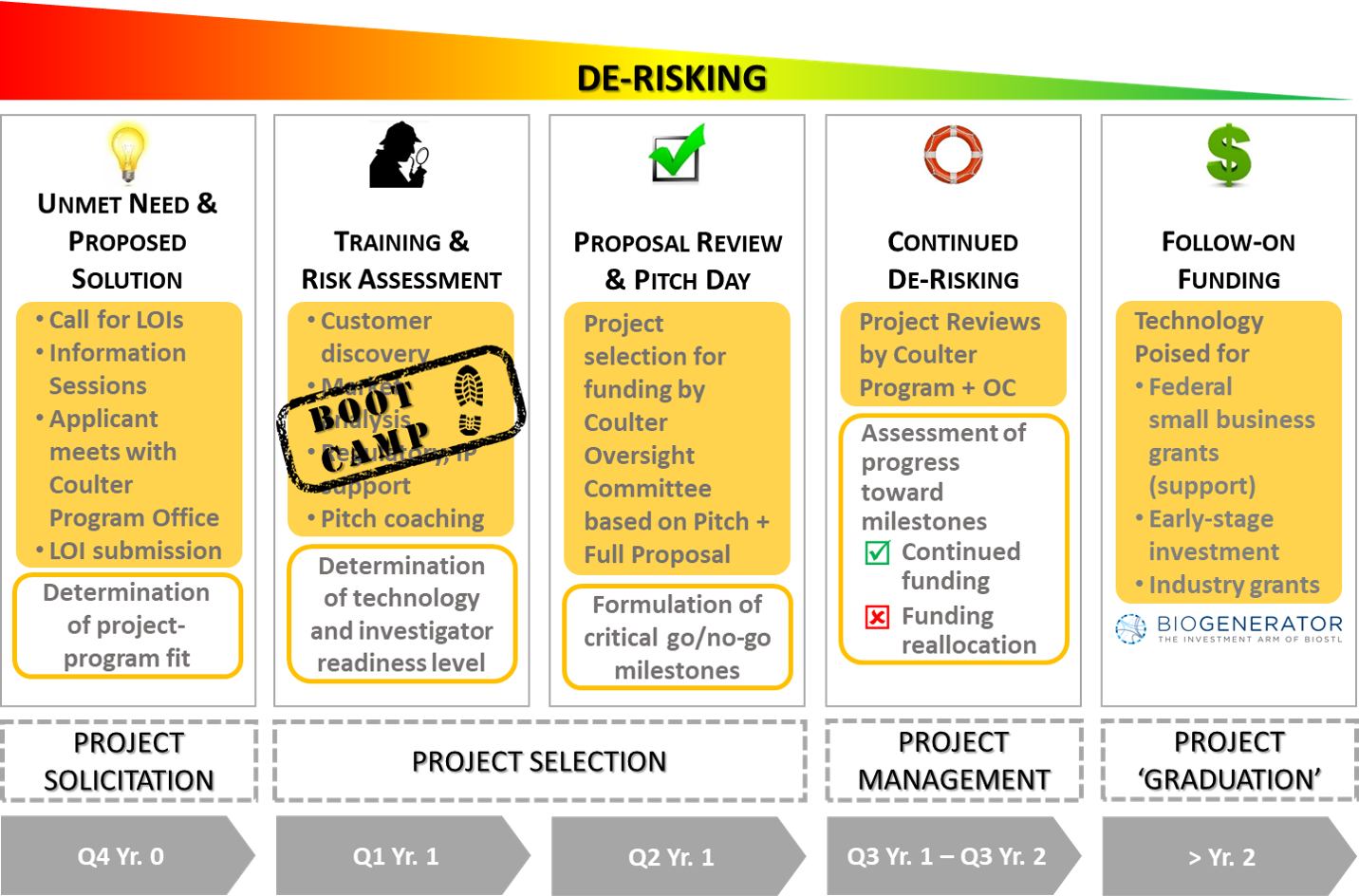
The multi-step application process includes submission of a Letter of Intent (LOI). LOIs are taken through a filtering process that includes a review by the Coulter Executive Committee. Applicants submitting LOIs that make it through the filtering process are invited to attend a commercialization Boot Camp that is now also offered in conjunction with MANGMT/ BIOL_EN 8200: Commercialization of Life Science Innovations. Teams that complete Boot Camp and submit a viable full proposal are given the opportunity to make a Pitch to the Coulter Oversight Committee (OC) for funding of the project. The OC makes the final decision on which projects are selected for funding. Funded projects are expected to start in Q3 of the funding year.
LOI Submission Guidelines: All investigators considering submission of a Letter of Intent (LOI) for a seed or full bridge award must meet with the Coulter Program Office before submitting an LOI. Please contact Jaya Ghosh (email: ghoshj@missouri.edu or call: 573-882-0522) to schedule a meeting.
Commercialization Boot Camp (now offered in conjunction with MANGMT/ BIOL_EN 8200: Commercialization of Life Science Innovations):
- Timeline: Q1 Yr. 1
- Objective: The primary objective of the Boot Camp is to help applicants answer two fundamental questions: 1) Does your envisioned product address a true unmet clinical need? AND 2) Is there a viable business opportunity? The second objective is to help applicants develop a robust proposal and pitch deck that best positions their idea for funding.
- Format: Invited speakers teach the basic principles of biomedical innovation and subject matter experts provide guidance on regulatory and reimbursement strategy, intellectual property and entrepreneurship. Applicants are also guided through four interactive home work sessions to identify stakeholders, define their value proposition and get-to-market strategy, develop their commercialization plan, and more. The Boot Camp and MANGMT/ BIOL_EN 8200 use the FROM LAB TO MARKET: The Process of Commercializing Medical Technologies and Biodesign Handbook, 1st Edition textbooks as guides.
- Boot Camp/Course Syllabus: Contact Jaya Ghosh for more details.
Full Proposal and Pitch Day:
- Timeline: Q2 Yr. 1
- Objective: Convince the OC that this grant will accelerate the transition of discovery originating from your academic research into a product or service that will improve human health by readying your technology for the next source of independent financing, such as the private sector, through a viable start-up company competitive for a small business award, or a licensing opportunity.
Coulter Funding
All PIs and collaborators on a funded project sign an Award Terms and Conditions Agreement prior to project start. Projects receive milestone-driven funding, and the Coulter Program Office meets with funded investigators on a monthly basis to track project progress. Investigators also meet with the OC on a regular basis to report progress toward milestones. Based on these updates, the OC makes go/no-go decisions on all funded projects. Non-progressing projects may be terminated.
No Cost Time Extension (NCTE) and Renewal: Coulter grants are awarded for a one-year period and may receive up to an additional six months in no cost time extension. Only seed projects are eligible for renewal. Renewal applications must include a comparison of milestones achieved versus milestones planned in the original submission. These applications are evaluated on a competitive basis with new applications.





